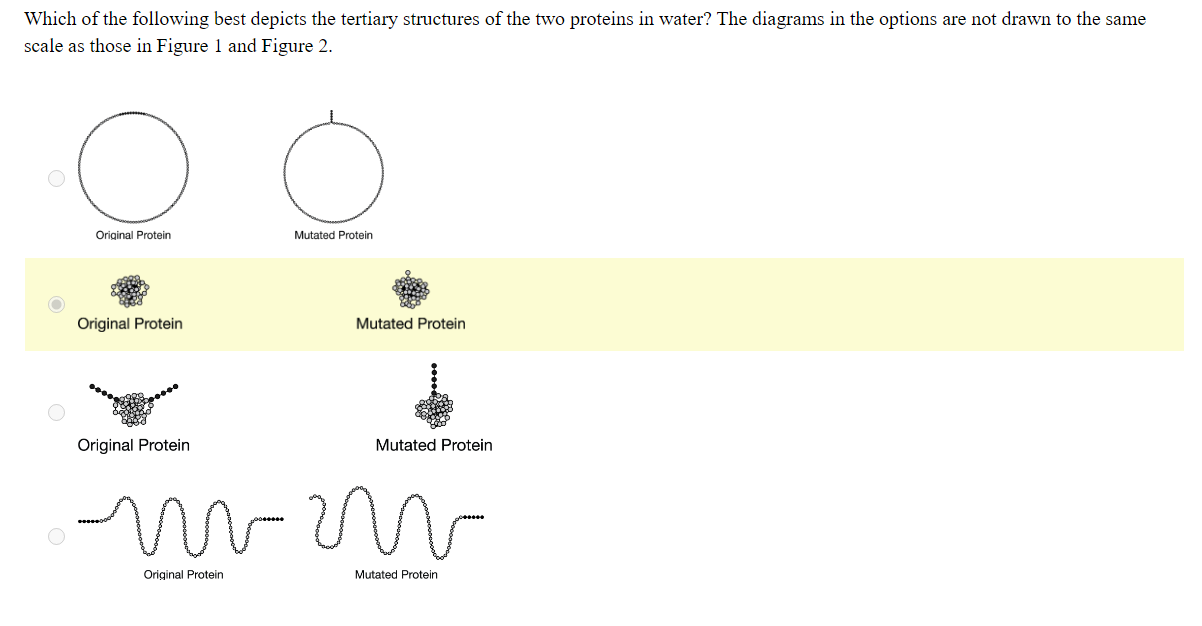
Hydrophobicity does not mean "love for hydrophobic atoms", but "fear of water".
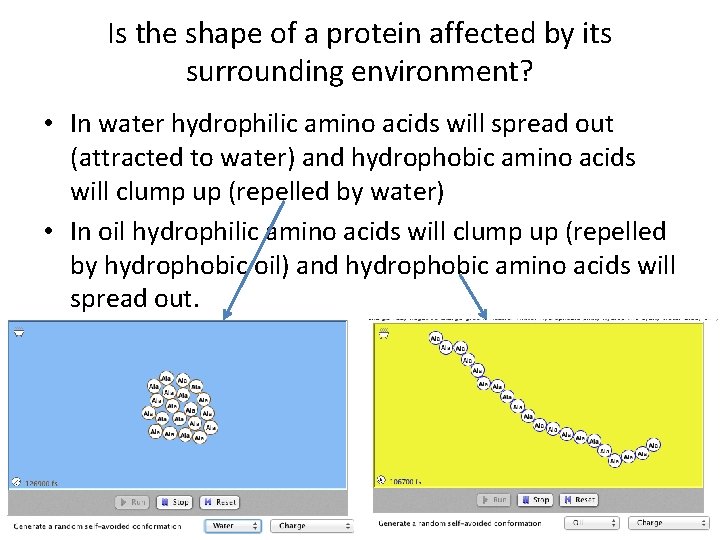
So, why do hydrophobic atoms want to sit close to each other inside a protein? For that we have to think again about the name of this effect: hydrophobicity. The force associated with this Van der Waals interaction is very weak at large interatomic distances and still weak when the atoms get closer. All atoms attract each other via the Van der Waals interaction. There doesn't exist such a thing as an 'attractive hydrophobic force'. Although this is a handy way to think about things, it is not correct. So, what is hydrophobicity? Many people describe it indirectly by saying things like "Hydrophobic residues attract each other via hydrophobic forces". Fortunately, most scales are reasonably similar, so for many practical purposes, it doesn't matter very much which scale you take. Which one you should use for your purpose depends on your question.

There are many hundreds of hydrophobicity scales in the literature (a hydrophobicity scale is a list of amino acids with corresponding hydrophobicity values). * From the atomic constitution of the sidechains * From the realtive distributions at the surface and in the core of proteins * From sidechain polarities as calculated by quantum chemical techniques * From octanol - water solubility differences Hydrophobicity has been measured or calculated in many different ways: Hydrophilic residues love water, and therefore like to sit at the outside of water-soluble proteins.īut what really is hydrophobicity? Well, that depends on the measurement. It is obvious that hydrophobic residues prefer to be in a non-aqueous environment, for example, inside a lipid bilayer. The words hydrophobic and hydrophilic literally mean 'afraid of water' and 'fond of water'. Hydrophobicity is not only the most important physico-chemical characteristic of amino acids, it is also the most poorly defined term.
